Leicester Microbial Sciences and Infectious Diseases Centre (LeMID)
Phage biology
Phage biology is the study of all aspects of bacteriophages (phages)
- Distribution
- Biochemistry
- Physiology
- Cell biology
- Ecology
- Evolution
- Therapeutic application
Bacteriophages are small viruses that infect bacteria. Some phages are lytic, meaning that they undergo a productive infection within a bacterial cell without integrating within the host genome (known as lysogeny), leading to the lysis and death of the bacterium. As such, there is a renewed effort to utilise phage, or cocktails of phage for the treatment of antibiotic resistant infection.
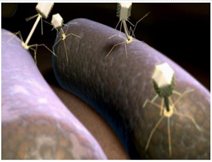
Bacteriophages have a high specificity for their bacterial hosts, rapidly killing their host and replicating themselves at the same time.
Generally, antibiotic resistance mechanisms in bacteria, have no role in bacterial defence against bacteriophages, meaning that antibiotic resistant bacteria can still be targeted by bacteriophages.
Additionally, biofilms, a key player in antibiotic resistant bacteria can be penetrated by bacteriophages that take advantage of a perfect amplification site, resulting in strong localised effects.
Professor Clokie's team studies the behavioural qualities and therapeutic possibilities against C. difficile. Studies include identification of and usage of bacteriophages against C. difficile and characterisation of C. difficile.
Phages in treatment
In 2016, funding was granted towards the ground-breaking research of Professor M. Clokie and Dr N Brown in the field of global health and development. As a bacteriophage biologist, Professor Clokie has an interest in bacteria that are difficult to treat by conventional antibiotics by developing phages as novel therapeutics.
C. difficile phage therapy
This would involve an oral dose of a specific phage preparation, the phage would then attach to the specific C. difficile cells preceding DNA entry, and these phage’s would be lytic. This would mean that a small dose would have a large effect as the phage DNA would replicate within the bacterium and be released to infect further bacterium. The treatment would also be limited by the number of C. difficile cells, therefore once all cells have been killed, there would be no platform for phage replication.
The advantages of Phage therapy for C.difficile
- Gut dysbiosis (disruption of gut microbiota) would not be aggravated due to the nature of phage-bacteria interaction
- Replication is self-limiting
- Delivery of phage to the colon is likely to present with little problems
- Limited number of strains of infective C. difficile means the phage profile is not extensive
- Commensal bacteria left intact due to specificity of phage to C. difficile
- Treatment is amplified locally, an attribute desirable when targeting biofilms
C. difficile
- Bacteria have developed multiple and diverse mechanisms to avoid and resist phage infection
- CRISPR/Cas system, a defence mechanism, evolved to resist bacteriophages
Clostridium difficile bacteriophages are effective anti-biofilm agents
Clostridium difficile causes antibiotic-induced diarrhoea and death in 10% of patients. Conventional antibiotics (e.g. vancomycin) cannot penetrate the protective extracellular polymetric substance (EPS) in biofilms and could result in treatment failure and recurrent C. difficile infection (CDI). We showed that pre-treatment of surfaces with bacteriophages (viruses of bacteria) (A) prevented biofilm formation and eliminated aggregated bacteria (B, C). In addition, bacteriophages lysed the EPS (D), mature biofilms (E) and free bacteria (F). Thus, bacteriophages are highly promising in the targeted eradication of CDI and are being developed to either supplement or replace antibiotics.
Phages in diagnostics
An important part of the research being carried out with bacteriophages at the University, is research investigating the use of phages to detect human bacterial infections, especially Lyme disease.

The main focus is isolation, characterisation and pre-clinical development of bacteriophages that target human pathogens, including tick-borne Borrelia strains (the causative agent of Lyme disease and Relapsing Fever). Recently, a phage-based diagnostics targeting Borrelia infection (Lyme disease) in collaboration with R.E.D Laboratories (Belgium), was invented by Dr Jinyu Shan and his team . This new test has been available as a commercial service since 1 July 2019.
The Chinese government plan to reduce antibiotic use in agriculture by 2020, therefore, phage therapy is being considered as a replacement for antibiotic use in farming. This is why the Genetics department has been working with a Chinese agriculture institute to introduce phage therapy to fight antimicrobial resistance in farming and irrigation.